Introduction
You cannot determine kon from an association binding measured at a single concentration of radioligand unless you can constrain koff to a constant value based on other experiments. The observed association rate depends on the association rate constant, the amount of ligand you used, and its dissociation rate constant. With one concentration of radioligand, the results are ambiguous. .
If you perform an association kinetic experiments with multiple radioligand concentration, you can globally fit the data to the association kinetic model to derive a single best-fit estimate for kon and one for koff.
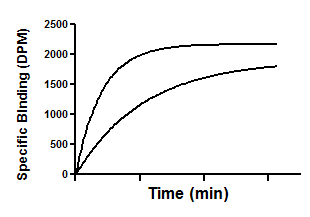
Shown below is an example of an association kinetic experiment conducted using two concentrations of radioligand. All other conditions (temperature, pH, etc.) were the same for both runs, of course. Times were entered into the X column, specific binding for one concentration of radioligand were entered into the first (A) Y column, and binding for the other concentration were entered into column B.
Step by step
Create an XY data table. Enter time in minutes into X, and total binding into Y. Enter binding at one concentration of radioligand into the column A, binding at another concentration into column B, etc. Enter the concentrations, in nM, into the column titles.
From the table of specific binding, click Analyze, choose nonlinear regression, choose the panel of Kinetics Binding equations, and choose Association - Two or more conc. of hot.
Model
Kd=Koff/Kon
L=Hotnm*1e-9
Kob=Kon*L+Koff
Occupancy=L/(L+Kd)
Ymax=Occupancy*Bmax
Y=Ymax*(1 - exp(-1*kob*X))
Interpret the parameters
Koff is the dissociation rate constant in min-1.
Kon is the association rate constant, in units of M-1 min-1.
Kd is the equilibrium dissociation constant, in Molar, computed as Koff/Kon
Bmax is the maximal binding at equilibrium, at maximal radioligand concentration, in the units you used to enter Y.
Notes
According to the law of mass action, the ratio of koff to kon is the Kd of receptor binding:
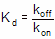
Compare the Kd calculated this way (from kinetic experiments) with the Kd determined from a saturation binding curve. If binding follows the law of mass action, the two Kd values should be indistinguishable.