Law of mass action
Kinetic binding experiments are used to determine the association and dissociation rate constants. All the analyses assume that binding follows the law of mass action:
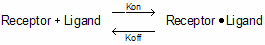
At any given time, the rate at which receptor-ligand complexes form is proportional to the radioligand concentration and the number of receptors still unoccupied. The rate of dissociation is proportional to the concentration of receptor-ligand complexes.
Rate of dissociation
A dissociation binding experiment measures the “off rate” for radioligand dissociating from the receptor. Initially ligand and receptor are allowed to bind, perhaps to equilibrium. At that point, you need to block further binding of radioligand to receptor (by adding an unlabeled drug or by dilution) so you can measure the rate of dissociation, which follows a one-phase exponential decay with a rate constant equal to the rate of radioligand dissociation.
Rate of association
In an association experiment, you add radioligand and measure specific binding at various times thereafter.
Binding increases over time until it plateaus. This plateau is not the same as the Bmax. The plateau in an association experiment depends on the concentration of radioligand used, while the Bmax is extrapolated to an infinite concentration of radioligand.
The rate at which binding increases is determined by three factors (as well as experimental conditions such as pH and temperature):
•The association rate constant, kon or k+1. This is what you are trying to determine.
•The concentration of radioligand. If you use more radioligand, the system equilibrates faster.
•The dissociation rate constant, koff or k-1. Some people are surprised to see that the observed rate of association depends in part on the dissociation rate constant. During the incubation, radioligand both binds to and dissociates from receptors. The system reaches equilibrium when the two rates are equal. The observed rate of association measures how long it takes to reach equilibrium. If the radioligand dissociates quickly from the receptor, equilibrium will be reached faster (but with less binding).
It is not possible, therefore, to fit a simple association experiment to determine the association rate constant unless you constrain the value of the dissociation rate constant, measure association kinetics with two or more concentrations of radioligand, or determine the rate of association and dissociation in one experiment.